TTP399
// TTP399
Type 1 Diabetes and Hypoglycemia
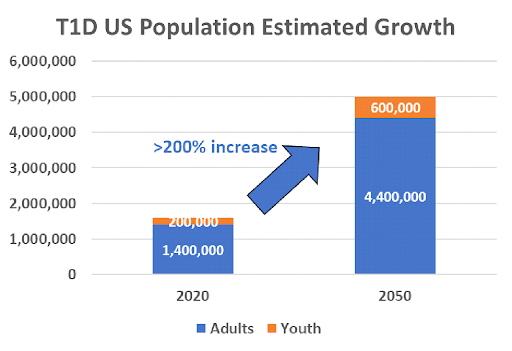
Type 1 Diabetes (T1D) is an autoimmune disease with no cure affecting over 1.6 million people in the United States. Adult-onset T1D has become more common than childhood-onset with more than half of all new cases diagnosed in adult patients. The patient population is expected to grow worldwide, with an estimated projection of 5 million patients in the U.S. by 2050.
Hypoglycemia is the most common acute complication in T1D, and hypoglycemic control continues to be an unmet need in the patient population.
Our Compound
TTP399 is a novel, oral, small molecule, liver selective glucokinase activator being developed as an adjunct therapy to insulin in patients with T1D. In a recent phase 2 study, TTP399 showed a 40% reduction in hypoglycemic episodes compared to placebo. In April 2021, the FDA granted Breakthrough Therapy designation to TTP399 for the treatment of T1D. This past October, vTv announced results of our mechanistic study of TTP399 in patients with T1D demonstrating no increased risk of ketoacidosis. TTP399 has now been tested in almost 600 subjects and demonstrated a good safety and tolerability profile.

Mechanism of Action
TTP399 is a Glucokinase (GK) Activator, a mechanism entirely distinct from the action of antidiabetic therapies currently on the market. TTP399 restores the normal function of the liver in the presence of high glucose: it traps glucose inside liver cells, promotes further glucose uptake for energy and storage, and keeps the liver in a “fed” state, thereby preventing ketone production. You can read more about the mechanism of action here.
// Clinical Trials
SimpliciT1 Phase 2 Study
TTP399 completed a phase 2 study in patients with T1D as an adjunctive treatment to insulin therapy. The study met its primary objective by demonstrating a statistically significant and clinically meaningful improvement to HbA1C, and treatment with TTP399 also showed a decrease in hypoglycemic episodes by 40%. TTP399 improved patients’ daily time in normal glycemic range with no reports of ketoacidosis during the study duration. Additionally, patients treated with TTP399 showed a reduction in their total daily mealtime bolus insulin dose.

You can read more about this study at ClinicalTrials.gov or read our article in Diabetes Care.
// Clinical Trials


// Clinical Trials
Type 2 Trial
TTP399 previously completed a 6-month phase 2 trial in 189 patients with type 2 diabetes where it achieved statistically significant reductions in HbA1c with negligible incidences of hypoglycemia and hyperlipidemia.